Wikisage, the free encyclopedia of the second generation, is digital heritage
Perampanel: Difference between revisions
Jump to navigation
Jump to search
mNo edit summary |
m (wLink) |
||
| Line 5: | Line 5: | ||
<ref>http://www.ema.europa.eu/docs/es_ES/document_library/EPAR_-_Product_Information/human/002434/WC500130815.pdf</ref> | <ref>http://www.ema.europa.eu/docs/es_ES/document_library/EPAR_-_Product_Information/human/002434/WC500130815.pdf</ref> | ||
Perampanel was efficacious and well tolerated, despite the observation that the patient population exhibited characteristics of highly refractory epilepsy | Perampanel was efficacious and well tolerated, despite the observation that the patient population exhibited characteristics of highly refractory [[epilepsy]] | ||
<ref>http://n.neurology.org/content/suppl/2016/04/10/WNL.0000000000001930.DC3/french_950.pdf</ref> | <ref>http://n.neurology.org/content/suppl/2016/04/10/WNL.0000000000001930.DC3/french_950.pdf</ref> | ||
| Line 17: | Line 17: | ||
{{wikidata|Q868658}} | {{wikidata|Q868658}} | ||
<references/> | <references/> | ||
[[Category:Anticonvulsant]] | [[Category:Anticonvulsant]] | ||
Revision as of 19:24, 10 January 2018
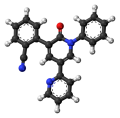
(Fycompa) is an AED [1] 5′-(2-Cyanphenyl)-1′-phenyl-2,3′-bipyridinyl-6′(1′H)-on
[2] Perampanel was efficacious and well tolerated, despite the observation that the patient population exhibited characteristics of highly refractory epilepsy