Wikisage, the free encyclopedia of the second generation, is digital heritage
Diacomit
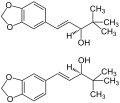
Diacomit contains 250 mg stiripentol[1][2]
In December 2001 the European Medicines Agency (EMA) granted stiripentol orphan drug status (designation number EU/3/01/071) for the treatment of severe myoclonic epilepsy of infancy (Dravet's syndrome). On 4 January 2007, the EMA granted the drug a marketing authorisation that is valid throughout the European Union
Dravet's syndrome
does not exist data for combined therapy both valproic acid or cloabazam[3]
ATC
http://www.whocc.no/atc_ddd_index/?code=N03AX17
- ↑ http://www.ema.europa.eu/docs/es_ES/document_library/EPAR_-_Product_Information/human/000664/WC500036518.pdf
- ↑ A new type of anticonvulsant, stiripentol. Pharmacological profile and neurochemical study
- ↑ Aras et al 2015