Wikisage, the free encyclopedia of the second generation, is digital heritage
Flibanserin: Difference between revisions
Jump to navigation
Jump to search
m (Tippfehler) |
(→Links: hide) |
||
| Line 6: | Line 6: | ||
==Links== | ==Links== | ||
http://jama.jamanetwork.com/article.aspx?articleid=2389384 Evaluation of Flibanserin Science and Advocacy at the FDA | [http://jama.jamanetwork.com/article.aspx?articleid=2389384 Evaluation of Flibanserin Science and Advocacy at the FDA] | ||
http://informahealthcare.com/doi/pdf/10.1517/14656566.2015.1064393 Why are there no FDA-approved treatments for female sexual dysfunction? | [http://informahealthcare.com/doi/pdf/10.1517/14656566.2015.1064393 Why are there no FDA-approved treatments for female sexual dysfunction?] | ||
<references/> | <references/> | ||
Revision as of 23:25, 24 August 2015
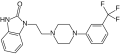
flibaserin is a new FDA aproved drug, it was formerly developed in Europe by Boeringer Ingelheim latter in US by Sprout Pharmaceutical due it refusal by Health Authority in 2013[1]Flibanserin is a post-synaptic 5-hydroxytryptamine (5-HT) 1A receptor agonist and 5-HT 2A antagonist that has been evaluated for indications of major depressive disorder (MDD) and for the treatment of HSDD in premenopausal women.[2]
Links
Evaluation of Flibanserin Science and Advocacy at the FDA
Why are there no FDA-approved treatments for female sexual dysfunction?