Wikisage, the free encyclopedia of the second generation, is digital heritage
Fabry disease: Difference between revisions
Jump to navigation
Jump to search
m (springer paper) |
mNo edit summary |
||
| Line 5: | Line 5: | ||
[[migalastat]] or 1-deoxygalactonojirimycin received an orphan drug approval | [[migalastat]] or 1-deoxygalactonojirimycin received an orphan drug approval | ||
<ref>https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/UCM616598.htm</ref> | <ref>https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/UCM616598.htm</ref> | ||
*improves diarrhea | |||
https://ojrd.biomedcentral.com/articles/10.1186/s13023-018-0813-7 | |||
[https://www.ncbi.nlm.nih.gov/pubmed/30064518 Deep characterization of the anti-drug antibodies developed in Fabry disease patients, a prospective analysis from the French multicenter cohort FFABRY] | [https://www.ncbi.nlm.nih.gov/pubmed/30064518 Deep characterization of the anti-drug antibodies developed in Fabry disease patients, a prospective analysis from the French multicenter cohort FFABRY] | ||
Revision as of 00:48, 11 August 2018
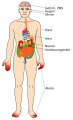
Fabry disease is a genetic condition involving lysosomal storage (E75.2)[1]is an X-linked disorder caused by alpha-galactosidase A deficiency [2]
migalastat or 1-deoxygalactonojirimycin received an orphan drug approval [3]
- improves diarrhea
https://ojrd.biomedcentral.com/articles/10.1186/s13023-018-0813-7 Deep characterization of the anti-drug antibodies developed in Fabry disease patients, a prospective analysis from the French multicenter cohort FFABRY