Wikisage, the free encyclopedia of the second generation, is digital heritage
Diacomit: Difference between revisions
Jump to navigation
Jump to search
No edit summary |
(v) |
||
| Line 4: | Line 4: | ||
In December 2001 the European Medicines Agency (EMA) granted stiripentol orphan drug status (designation number EU/3/01/071) for the treatment of severe myoclonic epilepsy of infancy (Dravet's syndrome). On 4 January 2007, the EMA granted the drug a marketing authorisation that is valid throughout the European Union | In December 2001 the European Medicines Agency (EMA) granted stiripentol orphan drug status (designation number EU/3/01/071) for the treatment of severe myoclonic epilepsy of infancy (Dravet's syndrome). On 4 January 2007, the EMA granted the drug a marketing authorisation that is valid throughout the European Union | ||
[[Dravet's | [[Dravet's syndrome]] | ||
does not exist data for combined therapy both valproic acid or cloabazam<ref>Aras et al 2015</ref> | does not exist data for combined therapy both valproic acid or cloabazam<ref>Aras et al 2015</ref> | ||
==ATC== | ==ATC== | ||
Revision as of 19:06, 31 March 2016
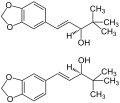
Diacomit contains 250 mg stiripentol[1]
In December 2001 the European Medicines Agency (EMA) granted stiripentol orphan drug status (designation number EU/3/01/071) for the treatment of severe myoclonic epilepsy of infancy (Dravet's syndrome). On 4 January 2007, the EMA granted the drug a marketing authorisation that is valid throughout the European Union
Dravet's syndrome
does not exist data for combined therapy both valproic acid or cloabazam[2]